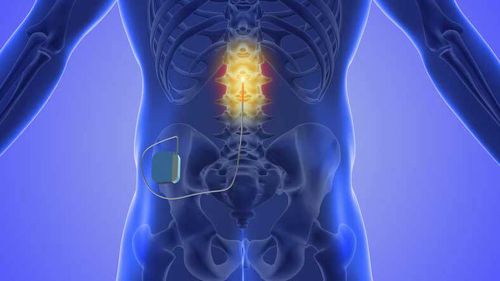
<div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>Australian doctors and health fund providers are calling for the use of spinal cord stimulators to be reconsidered after research revealed one-in-four people are having revision surgery for complications.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>Spinal cord stimulators have been used to treat chronic pain, including back pain, neck pain, nerve pain, and complex regional pain syndrome.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>The devices are made up of a small battery pack, which is implanted beneath the skin and attached to electrodes. Pulses of electricity are theoretically supposed to interfere with pain signals coming from the nerves.</span></div></div><div><div id="adspot-mobile-medium"></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><strong><span>READ MORE: </span></strong><a href="https://www.9news.com.au/national/federal-politics-parliament-returns-childcare-reforms-alleged-abuse/31d770b3-2124-4cc3-b8dd-b610c6ab772f"><strong><span>Urgent childcare reforms among priorities as parliament returns</span></strong></a></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>However, a study by leading Australian doctors, published in this week's </span><a href="https://onlinelibrary.wiley.com/doi/10.5694/mja2.70001?msockid=26b5191f26a46b9504f40a7f27b46a50"><span>Medical Journal of Australia</span></a><span>, found 23 per cent of patients fitted with the device will go on to need revision surgery for complications within three years.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>The median cost for the permanent device is nearly $56,000, with some patients' treatment adding up to more than $500,000.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>"Given the lack of supportive data for their efficacy, and a concerning harms profile, use of spinal cord stimulators for pain control should be reconsidered," the researchers wrote in the journal.</span></div></div><div><div class="OUTBRAIN" data-reactroot="" data-src="//www.9news.com.au/national/doctors-call-for-spinal-cord-stimulators-to-be-banned-as-study-reveals-major-problems-with-the-expensive-devices/168b670e-bf83-4d58-9f5a-24340e7d9f5c" data-widget-id="AR_5"></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>Lead author Dr Caitlin Jones, from the University of Sydney's Institute of Musculoskeletal Health, told </span><a href="https://www.smh.com.au/national/these-spinal-treatments-cost-more-than-50-000-a-new-study-found-they-do-not-work-20250718-p5mg1x.html" rel="" target="" title="The Sydney Morning Herald"><em><span>The Sydney Morning Herald</span></em></a><span> the risk posed by the devices was much greater than the likely benefits.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>"Patients should know they are taking on a substantial risk, that is exorbitantly expensive, with very little likelihood of any benefit."</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>Private Healthcare Australia, which represents health insurance funds and their 15 million members, has backed the researchers' calls for the use of the devices to be reconsidered.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>PHA CEO Dr Rachel David said while the Therapeutic Goods Administration (TGA) last year cancelled the registration of some spinal cord stimulators and imposed conditions on others, they were still available and being used on patients.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>"The regulatory action taken so far has been insufficient. Put simply, if these spinal cord stimulators were new technologies trying to get listed for Medicare funding in Australia, they would be deemed too dangerous," she said.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>"The Australian Government should ask the Medicare Services Advisory Committee to assess these devices and consider if they meet contemporary standards for listing. If not, the Medicare items and devices should be removed. This would protect patients from unnecessary harm and reduce wasteful expenditure for taxpayers and people with health insurance who are effectively being forced to fund this."</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>At least 90 per cent of spinal cord stimulators are inserted in the private health sector.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>"Health funds are committed to continuing to fund evidence-based treatments which have demonstrated safety and efficacy in the treatment of chronic pain," David said.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>The Medical Technology Association of Australia (MTAA), which represents device manufacturers has said the devices are safe and effective. </span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>Stimulators were "a demonstrably safe and effective alternative to long-term drug therapies, notably opioids", a spokesman told The Sydney Morning Herald.</span></div></div><div class="block-content"><div class="styles__Container-sc-1ylecsg-0 goULFa"><span>"[Stimulator] suppliers stand behind the safety, quality and efficacy of our SCS technologies that have demonstrated positive outcomes for patients, which are backed by robust clinical trial data and real-world evidence."</span></div></div>
Shock findings as doctors speak out on costly pain devices
Leave A Reply
Your email address will not be published.*